
What Is Week One Of Feeding?
I want to share my thoughts on what I believe is actually week one and when you should start feeding your plants hydroponic plant food. A lot of feeding charts don’t actually indicate...

I want to share my thoughts on what I believe is actually week one and when you should start feeding your plants hydroponic plant food. A lot of feeding charts don’t actually indicate...

For many growers, dealing with unresponsive seeds can be quite disheartening, especially if these seeds were a recent purchase. Don't worry; there are some helpful tricks that can come to...

Germinate seeds faster by using this germination tutorial by A-Grade Hydroponics. If you're local to our hydroponics shop in Melbourne cruise and and see some of the plant systems that...

Why buy seedlings or plants from a shop when you can germinate them yourself! By germinating seeds yourself you will save lots of money! When germinating seeds there are a...

What is cloning? Cloning within horticulture is the practice of taking a cut (cutting) from a desirable plant, propagating new root growth from the cutting which will be genetically identical to the original (mother)...

In a previous tutorial we discussed A-Grade Super-Cropping techniques, the many benefits to cropping, and how your plants can benefit from doing it. Following on from that, I want to talk...

Impatiens come in all shapes and sizes, they are a vigorous growing flowering plant. Flowers that form on impatiens plants are truly stunning, and come in a multitude of colours...

Plant cloning as it suggests, is copying an original, in this case a selected mother plant that is unique in characteristics that also expresses a specific set of expressions that...

Botanical name: Jasminum officinale L. Common name: White Jasmine Family: Oleaceae Aromatic compound: Eugenol, eugenyl acetate Ketones: Jasmonic acid, jasmolactone, methyl-di-hydro jasmonate, cis- and trans-ethyl jasmonate ~ “Before jumping...

Successful germination is on the mind of every gardener as we are always planning ahead, however some varieties can present a whole new learning curve just in their seed form....

Roots need the perfect environment to form. Ideally when rooting your cuttings or ‘clones’ we need to keep the medium or water temperature between 18.5 - 21.5 Celsius. The best...

The difference between regular lamps and proper grow lights can make or break your indoor garden. Learn more about more about what your indoor plants need.

Getting into and started with hydroponics can feel a bit like learning a new language. And the younger you are, the easier it is. Even then, between the different systems,...

The best way to get started with hydroponics is by having the right equipment from the beginning. Selecting a comprehensive hydroponic kit simplifies the process, ensuring you have all the...

Indoor growers are constantly trying to improve upon their previous results. As they become more experienced and confident, they might consider ways to optimise their indoor gardens for greater returns. The...

In the ever-evolving world of agriculture, hydroponic grow rooms have emerged as an integral tool for cultivating plants indoors. These indoor grow rooms effortlessly suit any growing styles; hand-feeding fabric...

For the last few decades the indoor horticulture and hydroponic industries have been reliant on using High Intensity Discharge (HID) and Fluorescent lighting technologies to illuminate their crops. These lighting...

Here is a neat guide from our friends at Apogee Instruments on the optimal light values for various crops. Many of our customers using full spectrum LED grow lights like...

Learn how to grow with LED full spectrum grow lights -A-Grade Hydroponics

What do all the Lighting Terminologies mean? Below is a glossary of terms commonly used to describe the way LED lighting is measured as well as some botanical stuff thats kind of...

Are you looking for the best hydroponic grow lights to help your plants thrive? In this blog post, we will discuss the different types of grow lights available and how...

This is a short tutorial on recommended hanging distances for the GRT Horticulture MLED-150 LED lighting bars. Introducing the GRT Horticulture MLED-150 The MLED-150 is an LED lighting bar...

Growing indoors is great fun but before you jump in and just buy any old light on eBay, read this informative post as it could save you a lot of...

Growing indoors is great fun, before you jump in and just buy any old light on eBay, read this informative post as it could save you a lot of money...

Hydroponic growing is lots of fun and very satisfying when things go well. With that in mind a lot of new and experienced gardeners have started to ask the question,...

When it comes to feeding your plants, there are many methods in which to do this. The most common systems for indoor growers rely on recirculation or wicking. These are...

One of the most important parts of running a healthy hydroponic system is getting your nutrient schedule right. In soil-based gardening, nutrients are slowly released over time. But hydroponics relies...

The 5 Best Hydroponic Nutrients for Premium Yields and Growth If you're looking for premium yields and growth in your hydroponic grow room or hydroponic garden, selecting the right nutrients...

In the world of gardening and horticulture, the health and vitality of plant roots are fundamental to overall plant growth. In most indoor grow room setups traditional containers, such as...

Have you ever wondered about Boron deficiency? It is actually more common than you think especially for those growing their favourite 'sacred' herbs and it's actually quite easy to avoid once...

How to feed plants indoors? Like anything living, energy is needed to fuel growth. Without food, organisms start to consume themselves, sacrificing health and food to sustain new growth which creates a host of...

So many choices, so many bottles but which hydroponic nutrients are the Best? With so many hydroponic nutrient brands on the market its easy to get pulled in with fancy...

A-Grade Hydroponics presents a handy Conversion Chart for those growers still using the truncheon ppm Meters. What is EC? Electrical conductivity or EC as it relates to hydroponics is a...

Copper deficiencies rarely occur in hydroponically-grown plants, especially when using a reputable hydroponic nutrient range. When a copper deficiency does occur it is usually mistaken for- and then treated- as...

Zinc is one of the eight essential micronutrients needed for healthy and productive plant life. Zinc plays multiple roles within plant cells and tissues, and even a minor zinc deficiency can...

Magnesium is the element responsible for a plant’s production of chlorophyll. A magnesium deficiency will impact a plant’s ability to photosynthesise, which in turn directly impacts the lush green colour...

Iron deficiency is quite a common problem in gardening, and we’re all bound to experience it sooner or later. Correctly identifying a plant deficiency can be difficult, as deficiencies sometimes...

See how Jase from A-Grade Hydro uses this bottle of liquid gold.

My latest obsession is the Monstera deliciosa. I love the almost prehistoric-sized leaves they produce; and the calm, almost hypnotising atmosphere created when grown and grouped indoors. Monstera deliciosa has...

Mycorrhiza is a mutual symbiotic association between a fungus and a plant. The term mycorrhiza refers to the symbiotic role of the fungus in the plant's root system. Endomycorrhizae is a...

“Get your plants off to a healthy start – a plant tonic like no other” Optimizing the growth of your plants, and finding the correct nutrient balance is essential. HY-GEN...

A-Grade's guide on feeding volumes and pot capacity Many customers ask us how much solution do they need to feed plants that are being grown under artificial lighting indoors and...

With winter in full swing, our phone has been going crazy with gardeners experiencing gardening issues. In short, the most common problem this time of year is the temperature once...

Potassium (K) is an essential nutrient for plant growth. It's classified as a macronutrient because plants take up large quantities of K during their life cycle. Potassium (K) is used in all fertilisers, potassium helps move water, nutrients and...

This product highlight will show off some of our favourite stocked bacterias and microbes designed to help the health and growth of your garden. Mammoth P Microbes by Growcentia...

Over the 7 years of trading at A-Grade Hydroponics, we've always loved and recommended HY-GEN's Humiboosta, making it our favourite product that works in almost all applications in conjunction with both...

Calcium (Cal) - Symbol: Ca Calcium is responsible for maintaining strong cell structure and vigour. High calcium levels are vital during the late growth stage, as additional calcium will ensure...

A great question, the amount we water/feed our plants is different for each growing medium used. The amount of water/food we use is subjective to the size of the pot...

The most commonly asked question: How much do I feed my plants? A great question, you need to feed your plants relative to their growth and needs. Feeding charts do...

This tutorial is provided by Hy-Gen's Founder and Chemist, Sandro. Optimizing the growth of your plants, and finding the correct nutrient level is essential. HY-GEN Australia, have developed a...

Plant nutrition is a broad topic that comes with many challenges. As a new gardener starting out, understanding the elements and how plants absorb/consume them is critical for long term success. Most growers,...

What is Nickels main function within a plant? Nickel (Ni) is a minor component of some plant enzymes, specifically urease which converts urea nitrogen into a useable form of ammonia...

Manganese (Mn), is an element, utilized as a micronutrient in plant nutrition. It is needed in trace amounts in early vegetation phase for successful germination and/or propagation. Mn deficiencies are quite...

Zinc Quick Facts. Zinc, is absorbed via the divalent Cation (Divalent; Valency of two include elements Ca & S) If a Zinc deficiency occurs, hormone production is decreased. Zinc...

Copper (Cu) is another interesting micro-element that doesn’t get much attention, it is a topic of great interest in agriculture as new understandings have been formed for recognizing deficiencies and...

Sulfur (S) is one of the nine essential minerals required by plants for growth. As sulfur problems are quite rare in comparison to other macro nutrient issues it can often...
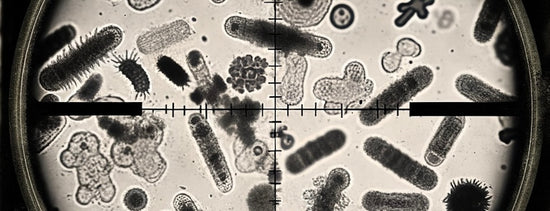
Colin Bell of Mammoth P talks about how unique microbial selection from plants play a large role in the productivity of specific crops. For those who are unaware of the...

Potassium (K) is an essential element that regulates growth and metabolism in plants. Potassium is also used in all fertilisers, primarily as a salt substitute and to help precipitate many...
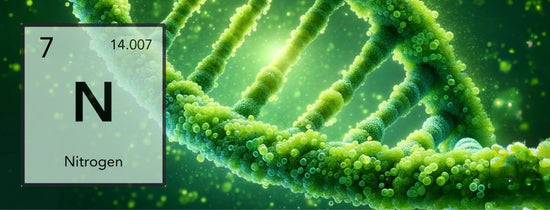
Nitrogen is one of the major elements needed by all life on earth (Nitrogen cycle) for plants nitrogen is one of the main elements responsible for chlorophyll production (Green pigment)...

Magnesium is a key element (core atom) in the production of chlorophyll, plants leaves/leaflets would not be able to form a ‘complete’ green pigment without magnesium’s presence. It has a...

Phosphorus is an essential element for plant growth, plants would not be able to grow/photosynthesise light without its presence. In humans, insufficient amounts of phosphorus in our bodies for an extended...

Like anything living, energy is needed to fuel growth. Without food, systems generally start to consume themselves creating a host of problems, each presenting a knock-on effect. Some example knock...
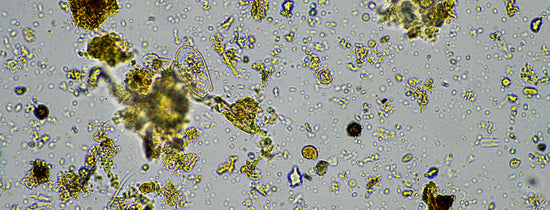
Introducing microbes to your plants is one of the most beneficial advancements you can give your plants health. Everyone who grows understand that plants have a few basic needs in...
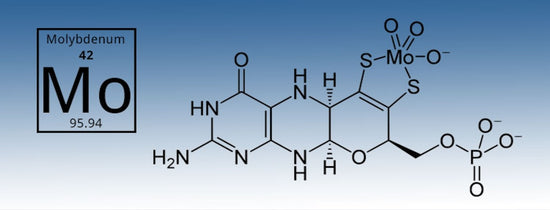
Molybdenum (Mo) is not only difficult to pronounce, (molyb-den-um) it is a needed element in gardening to accelerate nitrogen metabolism in plants. It does this by converting nitrates into nitrites...

Optimizing the growth of your plants, and finding the correct nutrient level is essential. Calcium-rich products play an important role in ensuring your garden thrives. Here, we look at just...

Maintaining a clean hydroponic grow room floor by removing any leaves and stems can reduce the likelihood of thrip infestations. Thrips thrive in these warm untidy areas especially amongst dying...

For some cannabis breeding has become a intricate combination of art and science; an unending quest to breed cannabis with superior genetics for potency, yield, resilience and stability. However, an invisible threat...

Fungus gnats are small flies very similar to fruit flies that belong to part of the Mycetophilidae and Sciaridae families. They feed on decaying organic matter, fungi and plant roots...

Fungus gnats are those small black annoying flies that seem to fly near your face at the most inconvenient times. They can very quickly infest your favourite plant pots if they...


Repeat after me: Prevention. Prevention. Prevention.

PRODUCT NAME: PURECROP1 PURPOSE: Insecticide, Fungicide & Biostimulant ORIGIN OF MANUFACTURE: USA CERTIFICATES: OMRI MAIN INGREDIENTS: Corn Oil, Soybean Oil PRODUCT CONFLICTS: None WHAT CAN I GROW? HERBS VEGETABLES FRUIT FLOWERS ORNAMENTALS...

Happy New Year A-Graders! We hope you all had the chance to catch up with family and friends over the break, and enjoy some time outdoors. The hot, wet summer...

An omnipresent disease. It is most commonly found on fiber crops but can also occur on plants forming natural medicines, notably Cannabis ruderalis. How Septoria affects crops Septoria leaf...

What is Neem Oil? Neem oil is a very popular pest preventative and direct treatment for most pest or mite infestations on plants. Neem oil is a vegetable oil extracted from...

Spider mites are little mites that are smaller than one mm in size and vary in a range of colours. Spider mites are a part of the Acari family Tetranychidae,...

Fungus Gnats. They are a part of the Mycetophilidae and Sciaridae super families. They feed on decaying organic matter, fungi and plant roots (which contain the nutrients you feed the plants)...

Scale bugs, of the Coccoidea family are roundish, oval disc looking bugs that have soft outer shells, and visually look light brown/orange. They attach themselves to the leaves and stems...

As the name indicates the virus is responsible for bizarre mosaic like patterning throughout the plant, causing a host of mRNA mutations like twisting, curling and in some cases severe...

This is a short article for those wondering the relative humidity (RH) requirements for each stage of a plants life cycle. Let’s start from the beginning of the plant cycle:...

Growing indoors in the hotter months can be quite tricky for growers not reliant on climate control systems to control this aspect of the grow themselves, have a read of this...

One crucial aspect that can significantly impact your plants health and yield is Vapour Pressure Deficit. In this blog, we'll explore what VPD is, why it matters, and how you...

This tutorial is designed for indoor gardeners growing plants in grow rooms and grow tents using hydroponic nutrients How does humidity affect my plants? The humidity in your grow room or grow tent...

This is a touched up version of a blog we did a few years ago where we explain the importance of maintaining certain temperatures during a plants lights off period. Why...

How does humidity effect my plants? Note: this tutorial is designed for growers using hydroponic nutrients. The humidity in the grow room will determine how your plants transpire. This directly...
It is quite cold outside at the moment, so in this article, we explain how important temperatures are for each stage of growing. Please note: The temperatures listed in this...

Humidity is an important environmental factor, so, too much or too little can make or break your grow, everyone has an opinion on the subject, and that’s all this is,...

It is quite cold outside at the moment, so in this article, we explain how important temperatures are for each stage of growing. Please note:The temperatures listed in this tutorial...

What is Vapour Pressure Deficit (VPD)? Simply put, VPD is the measurement of pressure in the atmosphere, which is measured and altered by temperature and humidity. Why does knowing my...

Carbon Dioxide is an odourless gas that is approx. twice the weight of oxygen. The main concern most indoor gardeners have when it comes to supplementing CO₂ is the worry...

Energy transformations are regulated by the laws of thermodynamics. Every single life form on earth and elsewhere, depend on energy transformations. Take our bodies for example, it transforms chemical energy...

In this article, we explain how important temperatures are for each stage of growing. What is the perfect temperature for the different stages of growth? Let’s start from the beginning:...

When growing plants indoors, as fun as the whole experience is there are also some unwanted issues that arise such as a ‘plant smell’ that most would want to control....

Growing auto flowering varieties has become quite popular amongst the new generation of hobby growers due to their fast turnaround and compact size, making them ideal for many new and...

Growing your own food is extremely rewarding! In this blog we take look at the basics of germinating seeds. Germinating your favourite plants from seed is a great way to start a...

Winter is in full swing and, that time where gardeners of all skill levels are preparing their indoor and outdoor gardens to grow food in for another bountiful season is...

Strawberries are one of the most popular fruits on Earth. They're also delicious and nutritious. But did you know they can be grown indoors using hydroponics? Read on to discover...

Hydroponics how to get started? Growing plants indoors is generally quite easy once the hydroponic growing equipment is in place and the environment is under control, for someone venturing into...

To start off this tutorial I’d like to list some of the most popular Day-Neutral crops grown both indoors & outdoors: Corn Strawberries Chilli Tomato Cucumber Cannabis Growing auto-flowering (day...

Tomato season has started again, this time I’ll be writing down a few revised tips from last year to help you get that heavy harvest, I found looking at the...

See Part One: How to grow and feed Chilli plants here If you’re reading this part of the Chilli blog then you have probably been growing your chilli seedlings...

Chilli are beautiful day-neutral plants; meaning they flower without regard to changes in light (photoperiod). As such, most chilli varieties can be grown both indoors and outdoors. The information below...

This is an updated and revised article as of 22/07/20 Recently at A-Grade Hydroponics we have observed a massive number of new growers and customers wanting to switch from their current...

This is a tutorial for growing in coco coir using hydroponic nutrients. Many growers often have trouble in the transition phases from seedling to their final pot. Many novice growers will...

Note: To be able to read PPFD or instantaneous PPFD you will require an accurate quantum meter to take the readings, such as the Apogee MQ-500. Most light meters measure...

To correctly calculate the VPD of your growing space you will need the following information: Ambient environmental temperature Relative humidity Leaf surface temperature Canopy level temperature First, find your saturated...

Nasturtiums are super easy to grow, and it wherever you grow them they have a wonderful screen of green effect! Nasturtiums also help deter many unwanted garden pests and they...

Growing herbs indoors is super easy and you can do it too! As you know, there are many varieties and hybrids of basil, none more sought after than the Greek...

We have grown a lot of crops before, but none is more enjoyable to grow than kale. From seed, kale germinate very fast in the right conditions, it is very...

Most gardeners love growing tomatoes and we are no different, tending to them (and any plant) can be a great way to clear the mind and is great stress relief!...

Growing herbs is heaps of fun, but so many people come to us as they are struggling to keep their home grown produce alive. Why do seedlings and established plants...

Drying-back is a deliberate, calculated and controlled method, with the objective of drying-back the coco coir just enough to stimulate vigorous root growth, but not leave the plant in a perished...

Gregor Mendel, is known in botany as the "father of modern genetics". Gregor chose to study variation in plants in his monastery's experimental garden (2ha > 4.9 acres). Terms like recessive...

The importance of quality botany or horticultural books in an industry shrouded in ‘Bro-science’ will hopefully provide you some relief. Books that help you with plant care and books written...

When growing plants, light provides the initial energy source for growth. The quantity and spectrum of the light is important for development of the plant, just as we measure the quantity...

The Cation Exchange Capacity (CEC) is a measure of a medium's ability to hold positively charged ions. This is an important property to manage in your coco/medium as it directly...

Breeding is lots of fun and if you’ve been growing for a while you would have encountered some amazing genetics and some, well, not so good ones. The name of...

Terpenes are a large and diverse class of organic compounds, produced by a variety of plants, particularly conifers, and by some insects. They often have a strong odour and may protect the plants that produce them by deterring...

In the last article I talked about general plant traits, in this I will explain why each of these is important and what to actually look for. Typically Sought After Traits...

When breeding any plant species there are certain outcomes that are considered desirable, these desirable outcomes are visibly shown in the form of traits and through successive breeding programs the...

Gregor Mendel is well known for being the first to observe multiple patterns of gene segregation for selected traits in peas and through some theoretical mapping, determine ratio outcomes for...
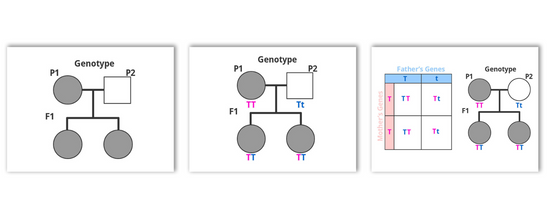
A pedigree looks like a family tree, it shows information about inherited traits throughout successive generations. A basic pedigree chart for a simple cross is quite easy to do: Circles...

Allele: The gene itself or allele, trait/s. Chemotype: Chemotypes are chemically distinct entities, with major differences occurring in secondary metabolites. Minimal genetic variance and little to no epigamic behaviour having...

Lots of you emailed in asking for us to start the discussion around breeding. So, let’s start with something basic that we can all wrap our heads around, Punnett squares!...

Triacontanol is an ultra-long-chain primary fatty alcohol (30:0) that is Triacontane in which one of the terminal methyl hydrogens is replaced by a hydroxy group. Common name: Triacontanol Chemical formula: C30H62O Chemical...
Have you ever wondered the mechanism behind how plants drink and eat? This tutorial is aimed to teach the basic mode of action. Translocation is the movement of materials from...

Hydrogen, oxygen and carbon are essential for the building blocks of life, they are considered non-mineral elements. They are also the three most used elements by plants. So, what do...
Note: I’m jumping ahead a little on this one. Sometimes we forget the importance of the little things and sometimes changes in our understanding of science and technology move forward. So, as I’m...

With traditional horticulture, the quality of the soil is an important factor. With hydroponics, it is the quality of the water. Sure, you'll likely be adding water to your hydroponics...

If you have been growing indoors under full spectrum led grow lights using a hydroponic system or in coco coir fabric grow pots, you should have a basic understanding of mixing hydroponic nutrients...

Growing hydroponically is a great way to grow crops, and offers a much greater return compared to conventional farming. However, just like conventional farming there are many variables to take...

Hydroponics is a form of agriculture that has gained immense popularity in recent years, especially for growing plants like herbs, vegetables and even weed. This method of growing allows plants...

Are you looking for a solution to improve plant growth and control bacteria and fungus in your hydroponics setup? Look no further than hydrogen peroxide! When used correctly, hydrogen peroxide...

Flushing is a technique used in between feeding weeks to ensure nutrition is available to the plant at all times, it is also used in the maturation (harvest) stage of a...

Having an understanding of how much food a plant requires helps massively as a gardener, however some don’t find it as easy, and forgoing the much-needed digital meters will only...

Note: This tutorial is an revision and update of a previous one from 2018 Algae are the oldest and most widely distributed form of plant life. They range...

Controlling your solution by creating good habits early, for strong, healthy plants and bountiful harvests, it’s not as difficult as it may seem, it’s just getting into the habit. So,...

Algae are the oldest and most widely distributed form of plant life. They range in size from the large ocean kelps to microscopic single cells, so small that a single drop of...

With summer coming up fast, an important topic that often gets overlooked that any grower who keeps a reservoir must deal with; reservoir temperature. For those who use true hydroponic...

A very important step in achieving excellence within the grow room! Most nutrient feed charts talk about weekly feeding so as your plants increase in volume, so does the nutrient...

Crop steering in coco coir is not very difficult to implement. If you understand the term relative to growth this tutorial will be much easier for you to understand, if not...

The Screen of Green (SCROG) method is a highly effective technique for cannabis cultivation in hydroponics. It involves training plants to grow through a screen or mesh, allowing for more...

Pruning and defoliation are two very different things but often confused with one another. PRUNING Pruning is the removal of any plant matter, usually with the intentions of manipulating growth...

To reduce unwanted energy flowing through to unwanted branches and stems which in turn redistributes to the remaining flowering sites. Don’t be fooled into thinking the four main branches technique...

Low Stress Training (LST) is a great technique used to increase overall yield, primarily used earlier on when plant stems and branches are flexible. Traditional grow vs LST grow If a plant is...

Last week we looked at some time saving perpetual gardening methods, this week we are going to show you how to perform a mainline on your plants. The process takes some...

It is no secret that training plants is the best way to stimulate growth and increase yield. BENEFITS OF SUPER CROPPING Yield & bud density Huge increases in flowering sites...

There are many techniques when it comes to plant training, let’s explore a few. When your plants are in a vegetive state they can become very large in a little...

Creating highly-productive plants doesn’t just happen naturally, it takes a lot of training, nutritional understanding and a healthy dose of intuition. With lots of misinformation available on how to top...

There are so many different techniques in regards to training your plants, all generally have the same end goal in mind, an increased yield. All of these techniques have been...

Coco coir has quickly become a popular choice among hydroponic growers due to its high cation exchange capacity (CEC) and favorable properties. This versatile growing medium is made from coconut...

Indoor plants are super addictive to collect and heaps of fun to grow and propagate. Imagine if there was a way we could maintain their environment all year round and...

Growing auto flowering varieties has become quite popular amongst the new generation of hobby growers due to their fast turnaround and compact size, making them ideal for many new and...

When plants perish or wilt, its not the end. Follow this guide by A-Grade Hydroponics to rescue your plants. With the right hydroponic equipment and know-how any wilting plant can...

A question I’ve been asked countless times. Tutorial time. So, why are your plants stretching? Well, when your plants stretch for light, this is called phototropism. This can be seen...

Lots of things can go wrong in any gardening space. Plants are living organisms, they breathe, eat, move and reproduce. Plants have special needs dictated by where they have originated...

What EC should I maintain in hydroponics? EC (Electrical conductivity) requirements change as the plant grows from seedling into a mature plant. When your plants are young they require less...

This is a quick tutorial on what to do (or not to do) if you experience purpling of the stems when transitioning from growth to flower. A lot of new...

When starting out a new gardening project it can take a few weeks to get everything right, some start of perfectly and others don’t so this tutorial is to help...

Our goal when growing plants is to achieve the best outcome possible, sometimes in this quest, we can make some mistakes. When such problems arise, take a deep breath and...

Hydroponics is mostly about keeping a few essentials in check. Here are the four essential hydroponic system elements that matter most.

Beginner-Friendly Plants for an Indoor Hydroponic Garden: Top Choices Your first decision when starting in hydroponics isn’t about nutrients or lighting. It’s about what you grow. Choose wisely and your...

What is the best LED light to grow with? Technology has come a long way and it is now far easier – and cheaper – to grow plants using artificial...

Should you turn your grow lights off at night? You might be wondering if you can ‘hack’ the system with LED grow lights. Theoretically, you leave them turned on 24/7...

How do hydroponic grow kits work? You don’t need a large outdoor area to grow plants anymore. Hydroponic growing is becoming increasingly popular and allows for the cultivating of plants...

Can plants get too much LED light? The old saying goes, ‘Everything in moderation’ – and that rings true when it comes to growing plants under LED lights. Like humans,...

Can any LED light be used as a grow light? We learn about photosynthesis in high school. This process begins with plants, algae and some bacteria converting light energy –...

How far should LED grow lights be from plants? When it comes to growing plants indoors, the correct placement of LED grow lights is critical. If your lights are too...

Well, Why Not? Nearly everything you buy from your local supermarkets is hydroponically grown and the prices are rising rapidly every year. So, we are encouraging people like yourself to join...

Hydroponics is a method of growing plants without soil. It is a more efficient way to provide water and nutrients to your plants. Soil provides nutrients, that must be broken...

An indoor hydroponic grow room can be set up just about anywhere, from a spare room to a garage or shed. There are just a few things that you need...

Good habits in hydroponics are as easy to form as bad ones; and one of the most annoying tasks is general maintenance of nutrient tanks and hydroponic systems. Most of these tasks are relatively...

What is EC? Electrical conductivity or EC is the measurement of the positive and negative ions in solution. To put it more simply EC indicates the strength of nutrients added to water and mixed....

At A-Grade Hydroponics we love meeting new growers; excited to start their passion for indoor gardening. For those used to traditional gardening methods, the change to liquid nutrients may be a wee...
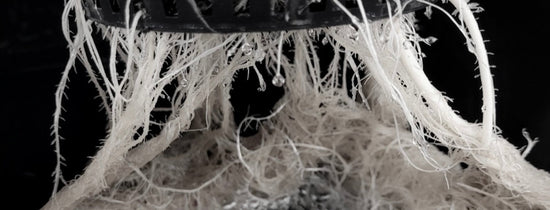
WHAT IS DEEP WATER CULTURE? Deep Water Culture - or ‘DWC’ - is a unique growing method involving the majority of the plant's root mass being permanently submerged in a nutrient solution. No...

Maintenance of reservoirs is relatively easy, by working smarter from the start you will find you have more time for the plants, and less time spent cleaning and fixing problems....

This tutorial will function as one of many guides to help people avoid making costly mistakes when using horticultural equipment and tools. Some of these rules will sound blatantly obvious and rather silly to mention, but never...

This week we want to discuss the importance of Nutrient Solutions. Your nutrient solution as you know is the food your plant needs as a requirement for growth. Growth however,...

Well, Why Not? Nearly everything you buy from your local supermarkets is hydroponically grown and the prices are rising rapidly every year. So, we are encouraging people like yourself to join...

Now we have a basic understanding of mixing our base nutrients, we can move on to checking the strength of our freshly made nutrient solution with an instrument called an...

When mixing nutrients, a lot of people ask us how to do it. Now the bottles do have instructions on, but there is still confusion. This quick tutorial will hopefully...